Recombivax-HB (Recombivax-HB)
1. Indications and Usage
RECOMBIVAX HB® [Hepatitis B Vaccine, Recombinant] is indicated for prevention of infection caused by all known subtypes of hepatitis B virus. RECOMBIVAX HB is approved for use in individuals of all ages. RECOMBIVAX HB Dialysis Formulation is approved for use in adult predialysis and dialysis patients 18 years of age and older.
2. Dosage and Administration
For intramuscular administration. See Section 2.2 for subcutaneous administration in persons with hemophilia.
RECOMBIVAX HB should be administered as soon as possible after being removed from refrigeration [see How Supplied/Storage and Handling (16)].
2.1 Dosage and Schedule
RECOMBIVAX HB:
Persons from birth through 19 years of age: A series of 3 doses (0.5 mL each) given on a 0-, 1-, and 6-month schedule.
Adolescents 11 through 15 years of age: A series of 3 doses (0.5 mL each) given on a 0-, 1-, and 6-month schedule or a series of 2 doses (1.0 mL each) on a 0- and 4- to 6-month schedule.
Persons 20 years of age and older: A series of 3 doses (1.0 mL each) given on a 0-, 1-, and 6-month schedule.
RECOMBIVAX HB Dialysis Formulation:
Adults on predialysis and dialysis: A series of 3 doses (1.0 mL each) given on a 0-, 1-, and 6-month schedule.
Table 1 summarizes the dose and formulation of RECOMBIVAX HB for specific populations, regardless of the risk of infection with hepatitis B virus.
| Group | Dose/Regimen |
|---|---|
| Infants*, Children and Adolescents 0-19 years of age (Pediatric/Adolescent Formulation) | 5 mcg (0.5 mL) 3 doses at 0, 1, and 6 months |
| Adolescents†
11 through 15 years of age (Adult Formulation) | 10 mcg‡ (1.0 mL) 2 doses at 0 and 4-6 months |
| Adults ≥20 years of age (Adult Formulation) | 10 mcg‡ (1.0 mL) 3 doses at 0, 1, and 6 months |
| Predialysis and Dialysis Patients§
(Dialysis Formulation) | 40 mcg (1.0 mL) 3 doses at 0, 1, and 6 months |
| |
2.2 Preparation and Administration
Shake the single-dose vial or single-dose prefilled syringe well to obtain a slightly opaque, white suspension before withdrawal and use. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Discard if the suspension does not appear homogeneous or if extraneous particulate matter remains or if discoloration is observed.
For single-dose vials, withdraw and administer entire dose of RECOMBIVAX HB intramuscularly using a sterile needle and syringe. Discard vial after use.
For single-dose prefilled syringes, securely attach a needle by twisting in a clockwise direction and administer dose of RECOMBIVAX HB intramuscularly. Discard syringe after use.
The deltoid muscle is the preferred site for intramuscular injection for adults, adolescents and children 1 year of age and older whose deltoid is large enough for intramuscular injection. The anterolateral aspect of the thigh is the preferred site for intramuscular injection for infants younger than 1 year of age. RECOMBIVAX HB should not be administered in the gluteal region, as injections given in the buttocks have resulted in lower seroconversion rates than expected.{2}
RECOMBIVAX HB may be administered subcutaneously to persons at risk for hemorrhage following intramuscular injections (e.g., hemophiliacs). However, hepatitis B vaccines are known to result in lower antibody response when administered subcutaneously.{3} Additionally, when other aluminum-adsorbed vaccines have been administered subcutaneously, an increased incidence of local reactions including subcutaneous nodules has been observed. Therefore, consider subcutaneous administration only in persons who are at risk of hemorrhage following intramuscular injections.
Do not administer intravenously or intradermally.
2.3 Known or Presumed Exposure to Hepatitis B Virus
Known or Presumed Exposure to HBsAg
Refer to recommendations of the Advisory Committee on Immunization Practices (ACIP) and to the package insert for hepatitis B immune globulin (HBIG) for management of persons with known or presumed exposure to the hepatitis B virus (e.g., neonates born of infected mothers or persons who experienced percutaneous or permucosal exposure to the virus). When recommended, administer RECOMBIVAX HB and HBIG intramuscularly at separate sites (e.g., opposite anterolateral thighs for exposed neonates) as soon as possible after exposure. Administer additional doses of RECOMBIVAX HB (to complete a vaccination series) in accordance with ACIP recommendations.
2.4 Booster Vaccinations
The duration of the protective effect of RECOMBIVAX HB in healthy vaccinees is unknown at present and the need for booster doses is not yet defined. The ACIP provides recommendations for use of a booster dose or revaccination series in previously vaccinated individuals with known or presumed exposure to Hepatitis B Virus.
Consider a booster dose or revaccination with RECOMBIVAX HB Dialysis Formulation (blue color code) in predialysis/dialysis patients if the anti-HBs level is less than 10 mIU/mL at 1 to 2 months after the third dose. Assess the need for a booster dose annually by antibody testing, and give a booster dose when the anti-HBs level declines to less than 10 mIU/mL.{3}
3. Dosage Forms and Strengths
RECOMBIVAX HB is a sterile suspension available in the following presentations:
- 0.5 mL (5 mcg) Pediatric/Adolescent Formulation single-dose vials and prefilled syringes
- 1 mL (10 mcg) Adult Formulation single-dose vials and prefilled syringes
RECOMBIVAX HB DIALYSIS FORMULATION is a sterile suspension available in the following presentation:
- 1 mL (40 mcg) single-dose vial [see Description (11) and How Supplied/Storage and Handling (16)]
4. Contraindications
Do not administer RECOMBIVAX HB to individuals with a history of severe allergic or hypersensitivity reactions (e.g., anaphylaxis) after a previous dose of any hepatitis B-containing vaccine or to any component of RECOMBIVAX HB, including yeast [see Description (11)].
5. Warnings and Precautions
5.1 Hypersensitivity to Latex
The vial stopper and the syringe plunger stopper and tip cap contain dry natural latex rubber, which may cause allergic reactions in latex-sensitive individuals.
5.2 Apnea in Premature Infants
Apnea following intramuscular vaccination has been observed in some infants born prematurely. Decisions about when to administer an intramuscular vaccine, including RECOMBIVAX HB, to infants born prematurely should be based on consideration of the individual infant's medical status and the potential benefits and possible risks of vaccination. For RECOMBIVAX HB, this assessment should include consideration of the mother's hepatitis B antigen status and the high probability of maternal transmission of hepatitis B virus to infants born to mothers who are HBsAg positive if vaccination is delayed.
5.3 Infants Weighing Less Than 2000 g
Hepatitis B vaccination should be delayed until 1 month of age or hospital discharge in infants weighing <2000 g if the mother is documented to be HBsAg negative at the time of the infant's birth. Infants weighing <2000 g born to HBsAg positive or HBsAg unknown mothers should receive vaccine and hepatitis B immune globulin (HBIG) in accordance with ACIP recommendations if HBsAg status cannot be determined{3} [see Dosage and Administration (2)].
5.4 Prevention and Management of Allergic Vaccine Reactions
Appropriate medical treatment and supervision must be available to manage possible anaphylactic reactions following administration [see Contraindications (4)].
5.5 Limitations of Vaccine Effectiveness
Hepatitis B virus has a long incubation period. RECOMBIVAX HB may not prevent hepatitis B infection in individuals who have an unrecognized hepatitis B infection at the time of vaccination. Additionally, vaccination with RECOMBIVAX HB may not protect all individuals.
6. Adverse Reactions
In healthy infants and children (up to 10 years of age), the most frequently reported systemic adverse reactions (>1% injections), in decreasing order of frequency, were irritability, fever, diarrhea, fatigue/weakness, diminished appetite, and rhinitis. In healthy adults, injection site reactions and systemic adverse reactions were reported following 17% and 15% of the injections, respectively.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a vaccine cannot be directly compared to rates in the clinical trials of another vaccine and may not reflect the rates observed in practice.
In three clinical studies, 434 doses of RECOMBIVAX HB, 5 mcg, were administered to 147 healthy infants and children (up to 10 years of age) who were monitored for 5 days after each dose. Injection site reactions and systemic adverse reactions were reported following 0.2% and 10.4% of the injections, respectively. The most frequently reported systemic adverse reactions (>1% injections), in decreasing order of frequency, were irritability, fever (≥101°F oral equivalent), diarrhea, fatigue/weakness, diminished appetite, and rhinitis.
In a study that compared the three-dose regimen (5 mcg) with the two-dose regimen (10 mcg) of RECOMBIVAX HB in adolescents, the overall frequency of adverse reactions was generally similar.
In a group of studies, 3258 doses of RECOMBIVAX HB, 10 mcg, were administered to 1252 healthy adults who were monitored for 5 days after each dose. Injection site reactions and systemic adverse reactions were reported following 17% and 15% of the injections, respectively. The following adverse reactions were reported:
Incidence Equal To or Greater Than 1% of Injections
GENERAL DISORDERS AND ADMINISTRATION SITE CONDITIONS
Injection site reactions consisting principally of soreness, and including pain, tenderness, pruritus, erythema, ecchymosis, swelling, warmth, nodule formation.
The most frequent systemic complaints include fatigue/weakness; headache; fever (≥100°F); malaise.
GASTROINTESTINAL DISORDERS
Nausea; diarrhea
RESPIRATORY, THORACIC AND MEDIASTINAL DISORDERS
Pharyngitis; upper respiratory infection
Incidence Less Than 1% of Injections
GENERAL DISORDERS AND ADMINISTRATION SITE CONDITIONS
Sweating; achiness; sensation of warmth; lightheadedness; chills; flushing
GASTROINTESTINAL DISORDERS
Vomiting; abdominal pains/cramps; dyspepsia; diminished appetite
RESPIRATORY, THORACIC AND MEDIASTINAL DISORDERS
Rhinitis; influenza; cough
NERVOUS SYSTEM DISORDERS
Vertigo/dizziness; paresthesia
SKIN AND SUBCUTANEOUS TISSUE DISORDERS
Pruritus; rash (non-specified); angioedema; urticaria
MUSCULOSKELETAL AND CONNECTIVE TISSUE DISORDERS
Arthralgia including monoarticular; myalgia; back pain; neck pain; shoulder pain; neck stiffness
BLOOD AND LYMPHATIC DISORDERS
Lymphadenopathy
PSYCHIATRIC DISORDERS
Insomnia/disturbed sleep
EAR AND LABYRINTH DISORDERS
Earache
RENAL AND URINARY DISORDERS
Dysuria
CARDIAC DISORDERS
Hypotension
6.2 Post-Marketing Experience
The following additional adverse reactions have been reported with use of the marketed vaccine. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to a vaccine exposure.
Immune System Disorders
Hypersensitivity reactions including anaphylactic/anaphylactoid reactions, bronchospasm, and urticaria have been reported within the first few hours after vaccination. An apparent hypersensitivity syndrome (serum-sickness-like) of delayed onset has been reported days to weeks after vaccination, including: arthralgia/arthritis (usually transient), fever, and dermatologic reactions such as urticaria, erythema multiforme, ecchymoses and erythema nodosum [see Warnings and Precautions (5.1)]. Autoimmune diseases including systemic lupus erythematosus (SLE), lupus-like syndrome, vasculitis, and polyarteritis nodosa have also been reported.
Gastrointestinal Disorders
Elevation of liver enzymes; constipation
Nervous System Disorders
Guillain-Barré syndrome; multiple sclerosis; exacerbation of multiple sclerosis; myelitis including transverse myelitis; seizure; febrile seizure; peripheral neuropathy including Bell's Palsy; radiculopathy; herpes zoster; migraine; muscle weakness; hypesthesia; encephalitis
Skin and Subcutaneous Disorders
Stevens-Johnson syndrome; alopecia; petechiae; eczema
Musculoskeletal and Connective Tissue Disorders
Arthritis
Pain in extremity
Blood and Lymphatic System Disorders
Increased erythrocyte sedimentation rate; thrombocytopenia
Psychiatric Disorders
Irritability; agitation; somnolence
Eye Disorders
Optic neuritis; tinnitus; conjunctivitis; visual disturbances; uveitis
Cardiac Disorders
Syncope; tachycardia
The following adverse reaction has been reported with another Hepatitis B Vaccine (Recombinant) but not with RECOMBIVAX HB: keratitis.
7. Drug Interactions
7.1 Concomitant Administration with Other Vaccines
Do not mix RECOMBIVAX HB with any other vaccine in the same syringe or vial. Use separate injection sites and syringes for each vaccine.
In clinical trials in children, RECOMBIVAX HB was concomitantly administered with one or more of the following US licensed vaccines: Diphtheria, Tetanus and whole cell Pertussis; oral Poliomyelitis vaccine; Measles, Mumps, and Rubella Virus Vaccine, Live; Haemophilus b Conjugate Vaccine (Meningococcal Protein Conjugate)] or a booster dose of Diphtheria, Tetanus, acellular Pertussis. Safety and immunogenicity were similar for concomitantly administered vaccines compared to separately administered vaccines.
In another clinical trial, a related HBsAg-containing product, Haemophilus b Conjugate (Meningococcal Protein Conjugate) and Hepatitis B (Recombinant) combination product (no longer licensed), was given concomitantly with eIPV (enhanced inactivated Poliovirus vaccine) or VARIVAX® [Varicella Virus Vaccine Live (Oka/Merck)], using separate sites and syringes for injectable vaccines. No serious vaccine-related adverse events were reported, and no impairment of immune response to these individually tested vaccine antigens was demonstrated.
The Haemophilus b Conjugate (Meningococcal Protein Conjugate) and Hepatitis B (Recombinant) combination product (no longer licensed) has also been administered concomitantly with the primary series of DTaP to a limited number of infants. No serious vaccine-related adverse events were reported.
7.2 Concomitant Administration with Immune Globulin
RECOMBIVAX HB may be administered concomitantly with HBIG. The first dose of RECOMBIVAX HB may be given at the same time as HBIG, but the injections should be administered at different sites.
7.3 Interference with Laboratory Tests
Hepatitis B surface antigen (HBsAg) derived from hepatitis B vaccines has been transiently detected in blood samples following vaccination. Serum HBsAg detection may not have diagnostic value within 28 days after receipt of a hepatitis B vaccine, including RECOMBIVAX HB.
8. Use in Specific Populations
8.1 Pregnancy
Risk Summary
All pregnancies have a risk of birth defect, loss, or other adverse outcomes. In the US general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4%, and 15% to 20%, respectively.
There are no adequate and well-controlled studies designed to evaluate RECOMBIVAX HB in pregnant women. Available post-approval data do not suggest an increased risk of miscarriage or major birth defects in women who received RECOMBIVAX HB during pregnancy.
Developmental toxicity studies have not been conducted with the vaccine in animals.
Data
Human Data
In post-licensure clinical studies of RECOMBIVAX HB, 26 pregnant women were inadvertently administered RECOMBIVAX HB following their last menstrual period. Among these pregnancies, after excluding elective terminations (n=3), there were 23 pregnancies with known outcomes all with exposure in the first trimester. Miscarriage was reported in 4 of 23 (17%) pregnancies and major birth defects were reported in 0 of 19 (0%) live births. The rates of miscarriage and major birth defects were consistent with estimated background rates.
Post-approval adverse reactions are reported voluntarily from a population of uncertain size. It is not always possible to reliably estimate their frequency or establish a causal relationship to the vaccine.
In prospectively reported spontaneous post-approval reports from 1986 to 2018, 105 women with known pregnancy outcomes were exposed to RECOMBIVAX HB during pregnancy following the last menstrual period. After excluding induced abortions (n=5), those with exposure in the third trimester (n=4), and those with an unknown exposure timing (n=6), there were 90 pregnancies with known outcomes with exposures in the first or second trimester. Miscarriage was reported for 7 of 90 (7.8%) pregnancies. Major birth defects were reported for 2 of 83 (2.4%) live born infants. The rates of miscarriage and major birth defects were consistent with estimated background rates.
8.2 Lactation
Risk Summary
It is not known whether RECOMBIVAX HB is excreted in human milk. Data are not available to assess the effects of RECOMBIVAX HB on the breastfed infant or on milk production/excretion.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for RECOMBIVAX HB and any potential adverse effects on the breastfed child from RECOMBIVAX HB or from the underlying maternal condition. For preventive vaccines, the underlying maternal condition is susceptibility to the disease prevented by the vaccine.
8.4 Pediatric Use
Safety and effectiveness of RECOMBIVAX HB have been established in all pediatric age groups. Maternally transferred antibodies do not interfere with the active immune response to the vaccine. [See Adverse Reactions (6.1) and Clinical Studies (14.1 and 14.2).] The safety and effectiveness of RECOMBIVAX HB Dialysis Formulation in children have not been established.
8.5 Geriatric Use
Clinical studies of RECOMBIVAX HB used for licensure did not include sufficient numbers of subjects 65 years of age and older to determine whether they respond differently from younger subjects. However, in later studies it has been shown that a diminished antibody response can be expected in persons older than 60 years of age.
11. Description
RECOMBIVAX HB Hepatitis B Vaccine (Recombinant) is a sterile suspension of non-infectious subunit viral vaccine derived from HBsAg produced in yeast cells. A portion of the hepatitis B virus gene, coding for HBsAg, is cloned into yeast, and the vaccine for hepatitis B is produced from cultures of this recombinant yeast strain according to methods developed in the Research Laboratories of Merck Sharp & Dohme LLC, Rahway, NJ, USA.
The antigen is harvested and purified from fermentation cultures of a recombinant strain of the yeast Saccharomyces cerevisiae containing the gene for the adw subtype of HBsAg. The fermentation process involves growth of Saccharomyces cerevisiae on a complex fermentation medium which consists of an extract of yeast, soy peptone, dextrose, amino acids and mineral salts. The HBsAg protein is released from the yeast cells by cell disruption and purified by a series of physical and chemical methods. The purified protein is treated in phosphate buffer with formaldehyde and then coprecipitated with alum (potassium aluminum sulfate) to form bulk vaccine adjuvanted with amorphous aluminum hydroxyphosphate sulfate. Each dose contains less than 1% yeast protein. The vaccine produced by the Merck Sharp & Dohme LLC, Rahway, NJ, USA method has been shown to be comparable to the plasma-derived vaccine in terms of animal potency (mouse, monkey, and chimpanzee) and protective efficacy (chimpanzee and human).
The vaccine against hepatitis B, prepared from recombinant yeast cultures, is free of association with human blood or blood products.
RECOMBIVAX HB Hepatitis B Vaccine (Recombinant) is supplied in three formulations. [See How Supplied/Storage and Handling (16).]
Pediatric/Adolescent Formulation (Without Preservative), 10 mcg/mL: each 0.5 mL dose contains 5 mcg of hepatitis B surface antigen.
Adult Formulation (Without Preservative), 10 mcg/mL: each 1 mL dose contains 10 mcg of hepatitis B surface antigen.
Dialysis Formulation (Without Preservative), 40 mcg/mL: each 1 mL dose contains 40 mcg of hepatitis B surface antigen.
All formulations contain approximately 0.5 mg of aluminum (provided as amorphous aluminum hydroxyphosphate sulfate, previously referred to as aluminum hydroxide) per mL of vaccine. In each formulation, hepatitis B surface antigen is adsorbed onto approximately 0.5 mg of aluminum (provided as amorphous aluminum hydroxyphosphate sulfate) per mL of vaccine. The vaccine contains <15 mcg/mL residual formaldehyde. The vaccine is of the adw subtype.
12. Clinical Pharmacology
12.1 Mechanism of Action
RECOMBIVAX HB has been shown to elicit antibodies to hepatitis B virus as measured by ELISA.
Antibody concentrations ≥10mIU/mL against HBsAg are recognized as conferring protection against hepatitis B infection.{2}
Infection with hepatitis B virus can have serious consequences including acute massive hepatic necrosis and chronic active hepatitis. Chronically infected persons are at increased risk for cirrhosis and hepatocellular carcinoma.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
RECOMBIVAX HB has not been evaluated for its carcinogenic or mutagenic potential, or its potential to impair fertility [see Use in Specific Populations (8)].
14. Clinical Studies
14.1 Efficacy in Neonates with Peripartum Exposure to Hepatitis B
The protective efficacy of three 5 mcg doses of RECOMBIVAX HB has been demonstrated in neonates born of mothers positive for both HBsAg and HBeAg (a core-associated antigenic complex which correlates with high infectivity). In a clinical study of infants who received one dose of HBIG at birth followed by the recommended three-dose regimen of RECOMBIVAX HB, chronic infection had not occurred in 96% of 130 infants after nine months of follow-up.{4} The estimated efficacy in prevention of chronic hepatitis B infection was 95% as compared to the infection rate in untreated historical controls.{5} Significantly fewer neonates became chronically infected when given one dose of HBIG at birth followed by the recommended three-dose regimen of RECOMBIVAX HB when compared to historical controls who received only a single dose of HBIG.{6} As demonstrated in the above study, HBIG, when administered simultaneously with RECOMBIVAX HB at separate body sites, did not interfere with the induction of protective antibodies against hepatitis B virus elicited by the vaccine.{6}
14.2 Immunogenicity of a Three-Dose Regimen in Healthy Infants, Children, and Adolescents
Three 5 mcg doses of RECOMBIVAX HB induced a protective level of antibody in 100% of 92 infants, 99% of 129 children, and in 99% of 112 adolescents [see Dosage and Administration (2.3)].
14.3 Immunogenicity of a Two-Dose Regimen in Healthy Adolescents 11 through 15 Years of Age
For adolescents (11 through 15 years of age), the immunogenicity of a two-dose regimen (10 mcg at 0 and 4-6 months) was compared with that of the standard three-dose regimen (5 mcg at 0, 1, and 6 months) in an open, randomized, multicenter study. The proportion of adolescents receiving the two-dose regimen who developed a protective level of antibody one month after the last dose (99% of 255 subjects) appears similar to that among adolescents who received the three-dose regimen (98% of 121 subjects). After adolescents (11 through 15 years of age) received the first 10-mcg dose of the two-dose regimen, the proportion who developed a protective level of antibody was approximately 72%.
14.4 Immunogenicity in Healthy Adults
Clinical studies have shown that RECOMBIVAX HB when injected into the deltoid muscle induced protective levels of antibody in 96% of 1213 healthy adults who received the recommended three-dose regimen. Antibody responses varied with age; a protective level of antibody was induced in 98% of 787 young adults 20-29 years of age, 94% of 249 adults 30-39 years of age and in 89% of 177 adults ≥40 years of age.
14.5 Efficacy and Immunogenicity in Specific Populations
Chronic Hepatitis C Infection
In one published study, the seroprotection rates in individuals with chronic hepatitis C virus (HCV) infection given the standard regimen of RECOMBIVAX HB was approximately 70%.{7} In a second published study of intravenous drug users given an accelerated schedule of RECOMBIVAX HB, infection with HCV did not affect the response to RECOMBIVAX HB.{8}
Predialysis and Dialysis Adult Patients
Predialysis and dialysis adult patients respond less well to hepatitis B vaccines than do healthy individuals; however, vaccination of adult patients early in the course of their renal disease produces higher seroconversion rates than vaccination after dialysis has been initiated.{9} In addition, the responses to these vaccines may be lower if the vaccine is administered as a buttock injection. When 40 mcg of Hepatitis B Vaccine (Recombinant), was administered in the deltoid muscle, 89% of 28 participants developed anti-HBs with 86% achieving levels ≥10 mIU/mL. However, when the same dosage of this vaccine was administered inappropriately either in the buttock or a combination of buttock and deltoid, 62% of 47 participants developed anti-HBs with 55% achieving levels of ≥10 mIU/mL.
15. References
- CDC. A Comprehensive Strategy to Eliminate Transmission of Hepatitis B Virus Infection in the United States. Recommendations of the Advisory Committee on Immunization Practices (ACIP) Part I: Immunization of Infants, Children and Adolescents. MMWR Recommendations and Reports 2005; 54(RR16): 1-23. Appendix C - Postexposure Prophylaxis of Persons with Discrete Identifiable Exposures to Hepatitis B Virus (HBV) and http://www.cdc.gov/hepatitis/hbv/pdfs/correctedtable4.pdf
- CDC. Suboptimal Response to Hepatitis B Vaccine given by Injection into the Buttock. MMWR Weekly Report 1985; 34: 105-8, 113.
- Centers for Disease Control and Prevention. A Comprehensive Immunization Strategy to Eliminate Transmission of Hepatitis B Virus Infection in the United States. Recommendations of the Advisory Committee on Immunization Practices (ACIP). Part 2: Immunization of Adults, MMWR 2006, 55(RR-16): 1-25.
- Stevens, C.E.; Taylor, P.E.; Tong, M.J., et al.: Prevention of Perinatal Hepatitis B Virus Infection with Hepatitis B Immune Globulin and Hepatitis B Vaccine, in Zuckerman, A.J. (ed.), "Viral Hepatitis and Liver Diseases", Alan R. Liss, 982-983, 1988.
- Stevens, C.E.; Taylor, P.E.; Tong, M.J., et al.: Yeast-Recombinant Hepatitis B Vaccine, Efficacy with Hepatitis B Immune Globulin in Prevention of Perinatal Hepatitis B Virus Transmission, JAMA 257(19): 2612-2616, 1987.
- Beasley, R.P.; Hwang, L.; Stevens, C.E.; Lin, C.; Hsieh, F.; Wang, K.; Sun, T.; Szmuness, W.: Efficacy of Hepatitis B Immune Globulin for Prevention of Perinatal Transmission of the Hepatitis B Virus Carrier State: Final Report of a Randomized Double-Blind, Placebo-Controlled Trial, Hepatology 3: 135-141, 1983.
- Wiedmann, M.; Liebert, U.G.; Oesen, U.; Porst, H.; Wiese, M.; Schroeder, S.; Halm, U.; Mossner, J.; Berr, F.: Decreased Immunogenicity of Recombinant Hepatitis B Vaccine in Chronic Hepatitis C, Hepatology, 31: 230-234, 2000.
- Minniti, F.; Baldo, V.; Trivello, R.; Bricolo, R.; Di Furia, L.; Renzulli, G.; Chiaramonte, M.: Response to HBV vaccine in Relation to anti-HCV and anti-HBc Positivity: a Study in Intravenous Drug Addicts, Vaccine, 17: 3083-3085, 1999.
- Recommendations of the Advisory Committee on Immunization Practices (ACIP): Hepatitis B Virus Infection: A Comprehensive Strategy to Eliminate Transmission in the United States, 1996 update, MMWR (draft January 13, 1996).
16. How Supplied/Storage and Handling
16.1 How Supplied
RECOMBIVAX HB (pediatric and adult) FORMULATION is available in single-dose vials and prefilled Luer-Lok® syringes.
RECOMBIVAX HB DIALYSIS FORMULATION is available in single-dose vials.
Pediatric/Adolescent Formulation (PRESERVATIVE FREE)
0.5 mL (5 mcg) in single-dose vials and prefilled Luer-Lok® syringes
NDC 0006-4981-00 – box of ten 0.5-mL single-dose vials
Color coded with a yellow cap and stripe on the vial labels and cartons and an orange banner on the vial labels and cartons
NDC 0006-4093-02 – carton of 10 prefilled single-dose Luer-Lok® syringes with tip caps
Color coded with a yellow plunger rod
Adult Formulation (PRESERVATIVE FREE)
1 mL (10mcg) in single-dose vials and prefilled Luer-Lok® syringes
NDC 0006-4995-00 – 1-mL single dose vial
Color coded with a green cap and stripe
NDC 0006-4995-41 – box of ten 1-mL single-dose vials
Color coded with a green cap and stripe
NDC 0006-4094-02 – carton of 10 pre-filled single-dose syringes with tip caps
Color coded with a green plunger rod
16.2 Storage and Handling
- Protect from light.
- Store vials and syringes at 2-8°C (36-46°F).
- Do not freeze since freezing destroys potency.
- RECOMBIVAX HB is stable at temperatures from 0° to 25° C (32° to 77°F) for 72 hours. These data are not recommendations for shipping or storage but may guide decisions for use in case of temporary temperature excursions.
17. Patient Counseling Information
Information for Vaccine Recipients and Parents/Guardians
- Inform the patient, parent or guardian of the potential benefits and risks associated with vaccination, as well as the importance of completing the immunization series.
- Question the vaccine recipient, parent or guardian about the occurrence of any symptoms and/or signs of adverse reaction after a previous dose of hepatitis B vaccine.
- Tell the patient, parent or guardian to report adverse events to the physician or clinic where the vaccine was administered.
- Prior to vaccination, give the patient, parent or guardian the Vaccine Information Statements which are required by the National Vaccine Injury Act of 1986. The materials are available free of charge at the Centers for Disease Control and Prevention (CDC) website (www.cdc.gov/vaccines).
- Tell the patient, parent or guardian that the United States Department of Health and Human Services has established a Vaccine Adverse Event Reporting System (VAERS) to accept all reports of suspected adverse events after the administration of any vaccine, including but not limited to the reporting of events by the National Childhood Vaccine Injury Act of 1986. The VAERS toll-free number is 1-800-822-7967. Reporting forms may also be obtained at the VAERS website at www.vaers.hhs.gov .
Spl Unclassified Section
Manufactured and Distributed by: Merck Sharp & Dohme LLC Rahway, NJ 07065, USA
For patent information: www.msd.com/research/patent
The trademarks depicted herein are owned by their respective companies.
Copyright © 1986-2023 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.All rights reserved.
uspi-v232-i-2304r443
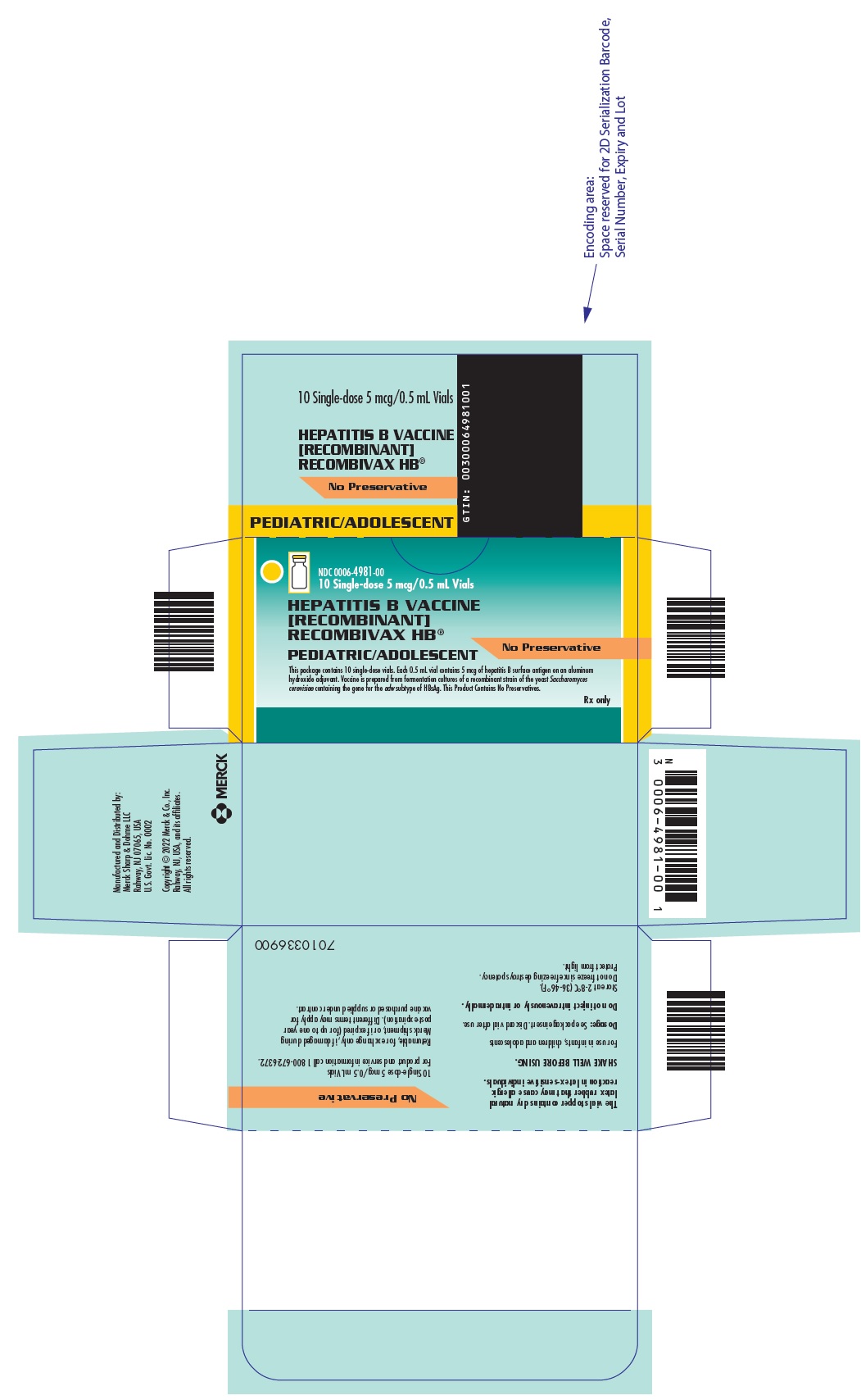
Package Label. Principal Display Panel
NDC 0006-4981-00 10 Single-dose 5 mcg/0.5 mL Vials
HEPATITIS B VACCINE [RECOMBINANT]RECOMBIVAX HB®
PEDIATRIC/ADOLESCENT
No Preservative
This package contains 10 single-dose vials. Each 0.5 mL vial contains 5 mcg of hepatitis B surface antigen on an aluminum hydroxide adjuvant. Vaccine is prepared from fermentation cultures of a recombinant strain of the yeast Saccharomyces cerevisiae containing the gene for the adw subtype of HBsAg. This Product Contains No Preservatives.
Rx only
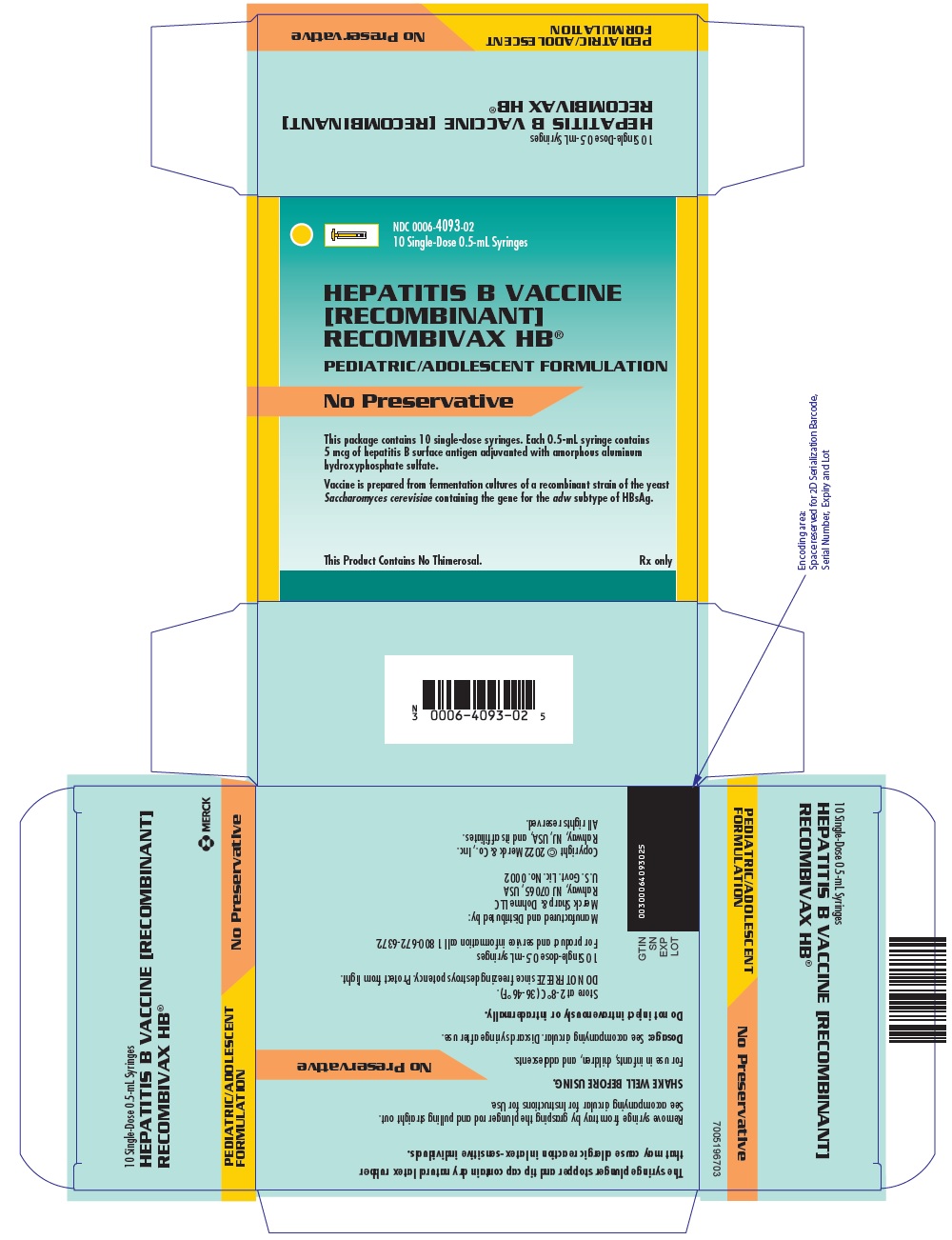
Package Label. Principal Display Panel
NDC 0006-4093-0210 Single-Dose 0.5-mL Syringes
HEPATITIS B VACCINE[RECOMBINANT]RECOMBIVAX HB®
PEDIATRIC/ADOLESCENT FORMULATION
No Preservative
This package contains 10 single-dose syringes. Each 0.5-mL syringe contains 5 mcg of hepatitis B surface antigen adjuvanted with amorphous aluminum hydroxyphosphate sulfate.
Vaccine is prepared from fermentation cultures of a recombinant strain of the yeast Saccharomyces cerevisiae containing the gene for the adw subtype of HBsAg.
This Product Contains No Thimerosal.Rx only
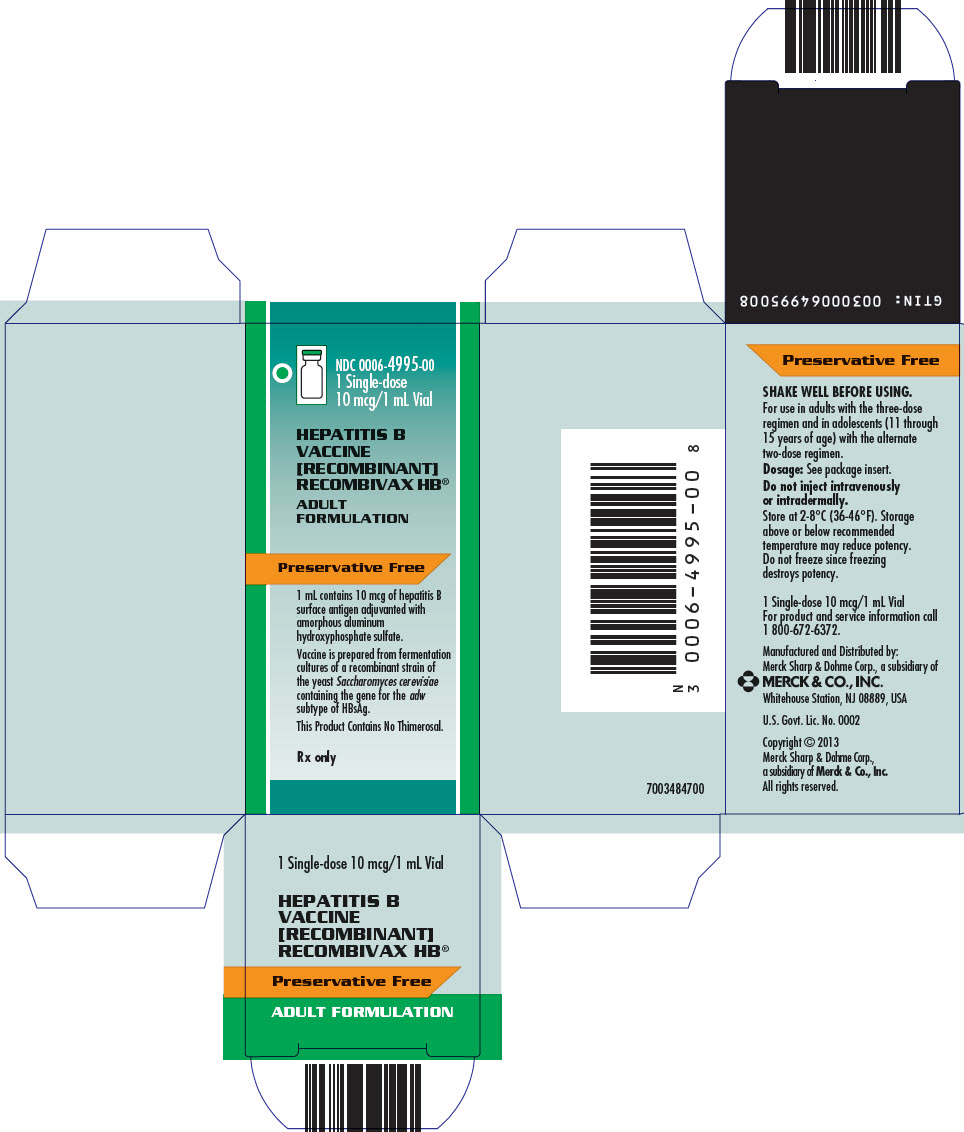
Package Label. Principal Display Panel
NDC 0006-4995-001 Single-dose10 mcg/1 mL Vial
HEPATITIS BVACCINE[RECOMBINANT]RECOMBIVAX HB®
ADULTFORMULATION
Preservative Free
1 mL contains 10 mcg of hepatitis B surface antigen adjuvanted with amorphous aluminum hydroxyphosphate sulfate.
Vaccine is prepared from fermentation cultures of a recombinant strain of the yeast Saccharomyces cerevisiae containing the gene for the adw subtype of HBsAg.
This Product Contains No Thimerosal.
Rx only
Package Label. Principal Display Panel
NDC 0006-4094-0210 Single-Dose 1.0-mL Syringes
HEPATITIS B VACCINE [RECOMBINANT]RECOMBIVAX HB®
ADULT FORMULATION
No Preservative
This package contains 10 single-dose syringes. Each 1.0-mL syringe contains 10 mcg of hepatitis B surface antigen adjuvanted with amorphous aluminum hydroxyphosphate sulfate.
Vaccine is prepared from fermentation cultures of a recombinant strain of the yeast Saccharomyces cerevisiae containing the gene for the adw subtype of HBsAg.
This Product Contains No Thimerosal.Rx only

Package Label. Principal Display Panel
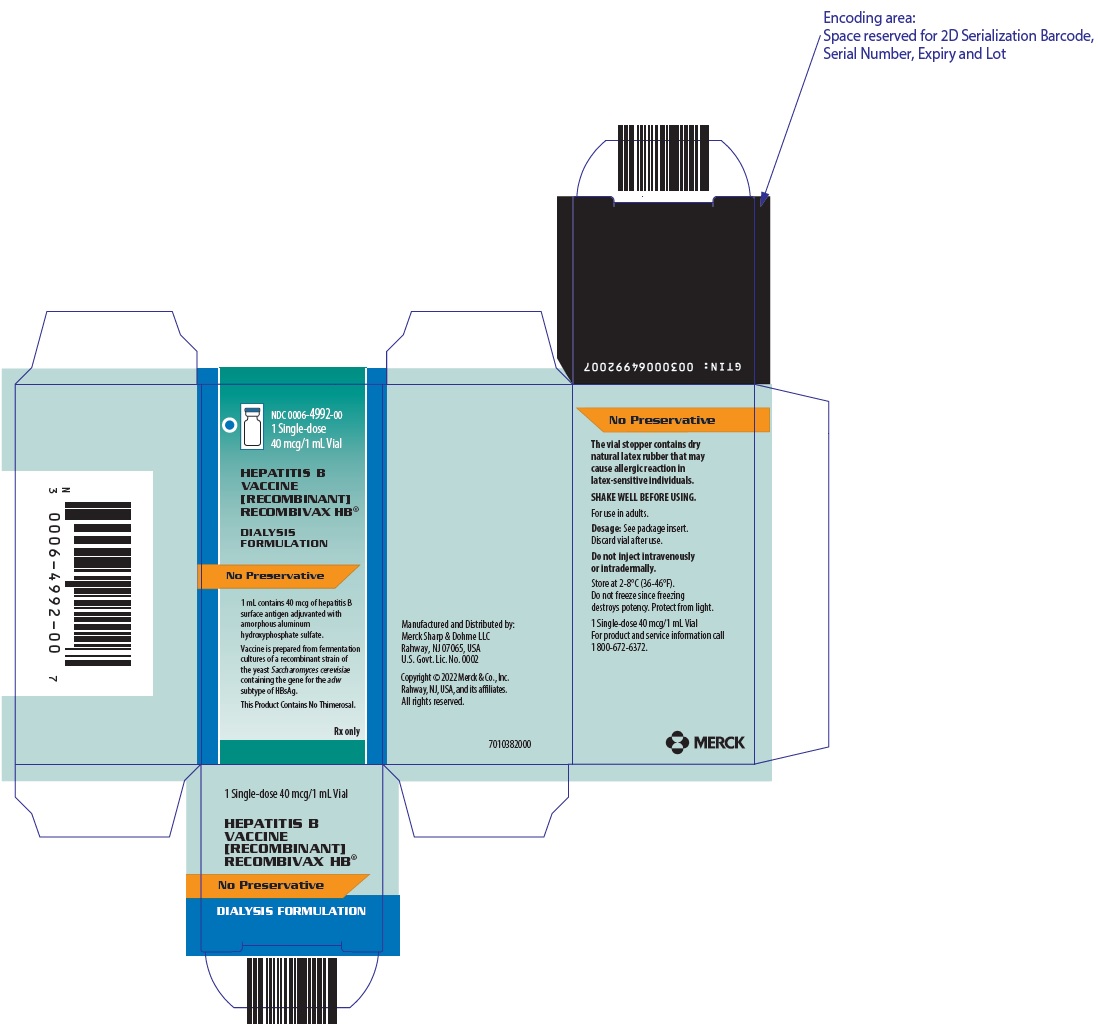
NDC 0006-4992-001 Single-dose40 mcg/1 mL Vial
HEPATITIS B VACCINE [RECOMBINANT]RECOMBIVAX HB®
DIALYSIS FORMULATION
No Preservative
1 mL contains 40 mcg of hepatitis B surface antigen adjuvanted with amorphous aluminum hydroxyphosphate sulfate.
Vaccine is prepared from fermentation cultures of a recombinant strain of the yeast Saccharomyces cerevisiae containing the gene for the adw subtype of HBsAg.
This Product Contains No Thimerosal.
Rx only
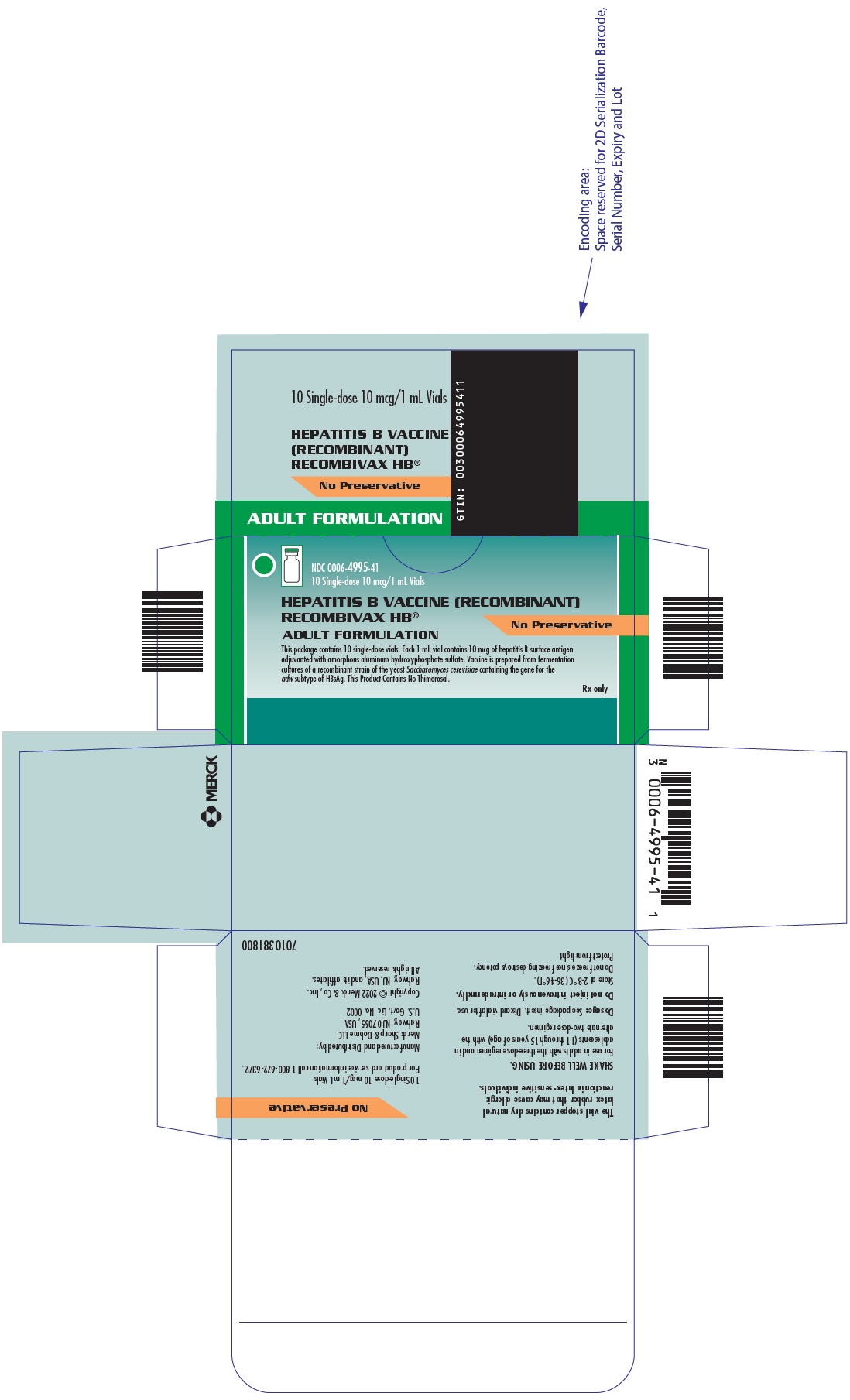
Package Label. Principal Display Panel
NDC 0006-4995-4110 Single-dose 10 mcg/1 mL Vials
HEPATITIS B VACCINE (RECOMBINANT)RECOMBIVAX HB®
No Preservative
ADULT FORMULATION
This package contains 10 single-dose vials. Each 1 mL vial contains 10 mcg of hepatitis B surface antigen adjuvanted with amorphous aluminum hydroxyphosphate sulfate. Vaccine is prepared from fermentation cultures of a recombinant strain of the yeast Saccharomyces cerevisiae containing the gene for the adw subtype of HBsAg. This Product Contains No Thimerosal.
Rx only